
부메뉴
Research
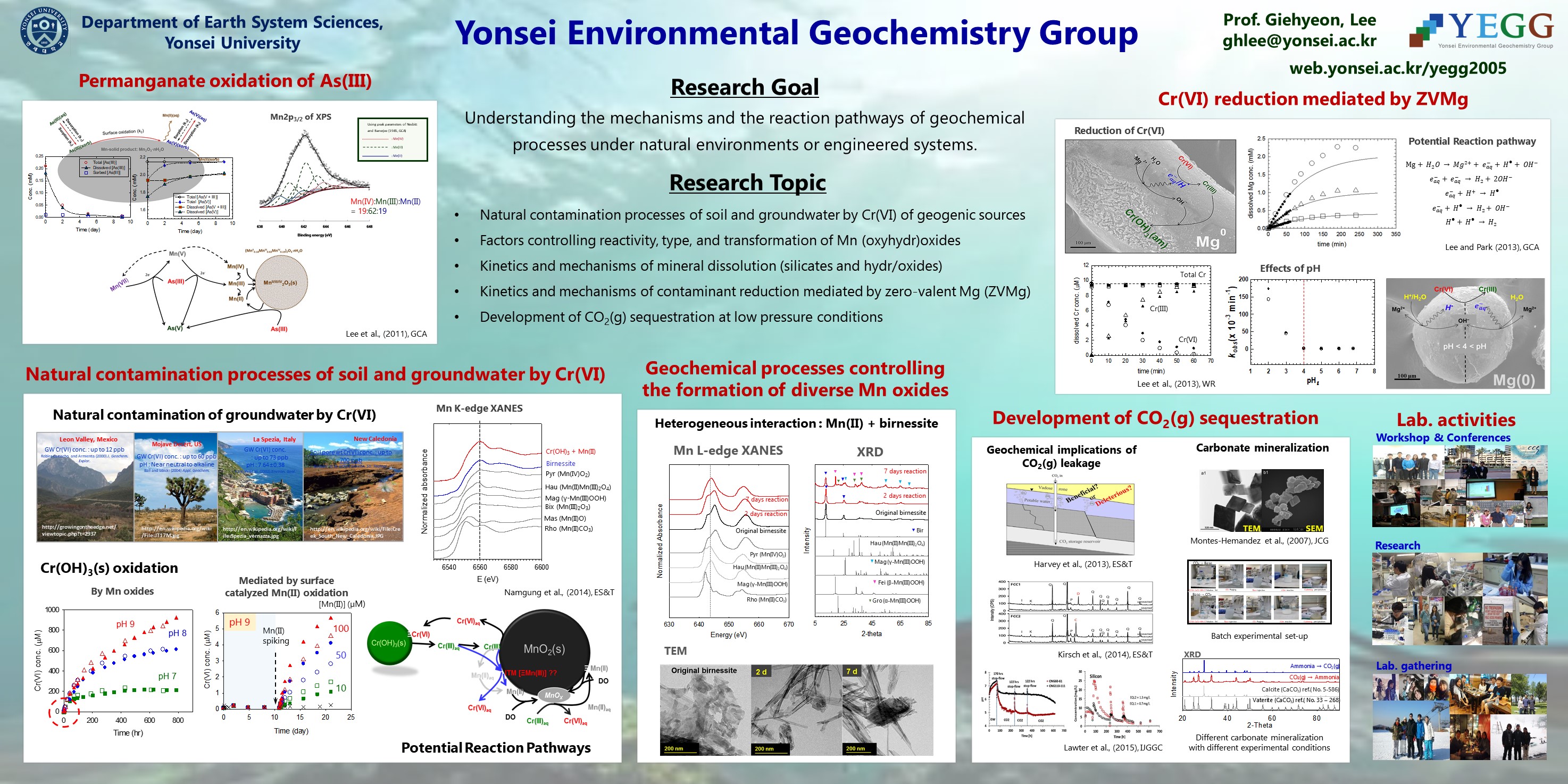
Permanganate oxidation of arsenic(III)
-
- Arsenic (As) is deleterious to human health, inorganic As(III) and As(V) are prevalent in natural water. As(III) is known to be more toxic and more mobile than As(V). Accordingly, the processes of removing As from drinking water have been conventionally utilized for the pre-oxidation of As(III) to As(V), followed by a process of coprecipitation or adsorption. Permanganate (MnO4-) has widely been used as an effective oxidant for drinking water treatment systems, as well as for in-situ treatment of groundwater. The purpose of this study is to determine the mechanism of As(III) oxidation by permanganate, therefore we determined the stoichiometry of As(III) oxidation by permanganate, characterized the Mn solid product from the reaction, and examined the feasibility of secondary heterogeneous oxidation of As(III). We also discussed the potential reaction pathway of As(III) oxidation by permanganate.
-
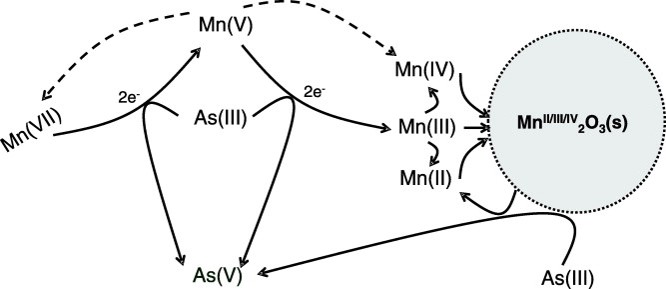
Natural contamination of soil and groundwater by Cr(VI) of natural origin
-
- In preparation.
-
ZVMg
-
- Recently, zero-valent magnesium (ZVMg) has been regarded as an effective reductant of environmental contaminants for potential applications in the remediation processes. Utilization of ZVMg for the treatment of contaminants would have several advantages; (1) ZVMg (E0SHE = -2.37 V) has the highest reduction potential among zero-valent metals; (2) the nuisance effect of surface passivation may be insignificant due to the substantially high solubility of ZVMg corrosion products; and (3) ZVMg can be applied under both aerobic and anaerobic conditions. Despite the potential advantages of ZVMg, research of removing contaminants with ZVMg is uncommon. Therefore, we are examining the reactivity of ZVMg in aqueous solution as a strong and efficient reducing agent of various contaminants and trying to elucidate the corresponding reaction pathways and mechanisms of the contaminant removal processes. In addition, we have been investigating the feasibility of practical application of ZVMg wasted from industrial manufacturing processes using metallic Mg and Mg alloy.
-

Geochemical processes controlling the formation of diverse Mn oxides
-
- Manganese (oxyhydr)oxides (hereafter referred to as ‘Mn oxides’) are widely distributed in most geological environments and found in more than 30 different types. In particular, each type of Mn oxides is characterized by distinct reactivity depending on its physicochemical properties (i.e., crystal structure and the valence state of structural Mn). In the natural environment, Mn oxides may play a key role in controlling the fate and transport of various inorganic and organic substances by participating in complex (bio)geochemical processes. It is well known that during (bio)geochemical processes including homogeneous, surface catalyzed, or microbial Mn(II) oxidation processes, formation and phase transformation of numerous types of Mn oxides may occur. Here, we are attempting to experimentally determine abiotic geochemical processes and factors controlling the pathways of the formation and the phase transformation of various Mn oxides which still have not been clearly understood yet.

Adsorption behavior of Mn(II)
-
- In preparation

